Scientists believe they have developed an affordable test that will increase a woman's chance of getting pregnant through IVF by 70 to 80%.
The test, which costs just £200, looks for abnormal levels of cells called mitochondrial DNA in the IVF embryos.
Mitochondrial DNA typically converts energy from food into a form that cells can use. However, according to scientists, a high level of mitochondrial DNA "indicates that the embryo is struggling".
Once the test has identified the defective embryos, they can be removed before IVF implantation occurs.
The scientists state that as the health of the embryo is one of the most important factors contributing to the success of IVF treatment, they believe the test will dramatically increase a woman's chances of getting pregnant.
Dr Elpida Fragouli, who led the research at Reprogenetics said: "Based on our findings we have devised a test whereby a small number of cells, carefully removed from an embryo, can be measured for the amount of mitochondrial DNA present.
"This will help guide doctors to the IVF embryos with the greatest chance of producing a viable pregnancy."
The scientists in the study noted high levels of mitochondrial DNA in embryos tend to increase as a woman ages.
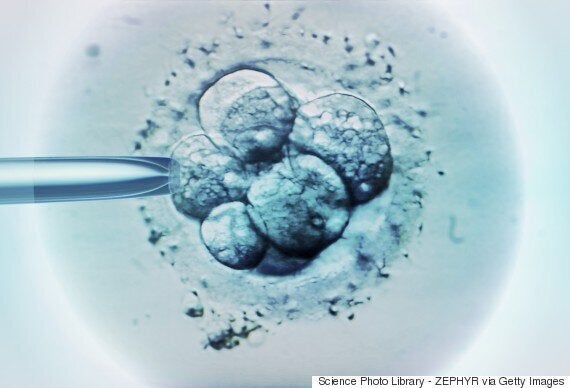
IVF doctors currently study embryos under a microscope in order to identify those with the best chance of implanting in the womb - they believe this gives a 60-65% success rate.
Dr Amin Gorgy, fertility consultant and co-director at The Fertility & Gynaecology Academy told HuffPost UK Parents: "This could be the answer to successful IVF in the future."
"The embryos that implant successfully have lower mitochondrial DNA. Those with a higher amount are called 'struggling embryos' because they try to compensate the high levels by using more energy and are not successful.
"The test is an additional step to further increase the implantation ratio."
Explaining the link between mitochondrial DNA and a woman's fertility, Prof Dagan Wells of the NIHR Oxford Biomedical Research Centre said, according to The Telegraph: "Embryos with elevated levels of mitochondrial DNA never seem to implant. It's something to do with energy production that isn't quite right. It's given us a new bio marker.
"You have to take a small number of cells five days after the egg has become fertilised. It is likely to raise implanting to around 70 to 80%."
The test is already being used in the US and is currently awaiting approval by the Human Fertilisation and Embryology Authority (HFEA) in the UK. This is hoped to be within six months.
The research was presented at the American Society for Reproductive Medicine annual meeting in Baltimore.