Around 80 years ago it was theorised that with enough pressure, you could in fact transform hydrogen into a metal.
Well now two scientists at Harvard University claim to have turned that theory into reality and created metallic hydrogen.
Hydrogen in its metallic form has some truly incredible properties. For starters it could be metastable, which means that once created it would retain its state even if you brought it back to ambient conditions. It is also a zero resistance superconductor, which means it could revolutionise the fields of technology and electricity storage.
Finally, it’s unbelievably powerful as a fuel source. NASA has long sought to create hydrogen in solid form knowing that it could allow them to achieve levels of thrust not yet seen by humanity.
The problem has always been that to create such a substance you have to exert astronomical quantities of pressure on hydrogen.
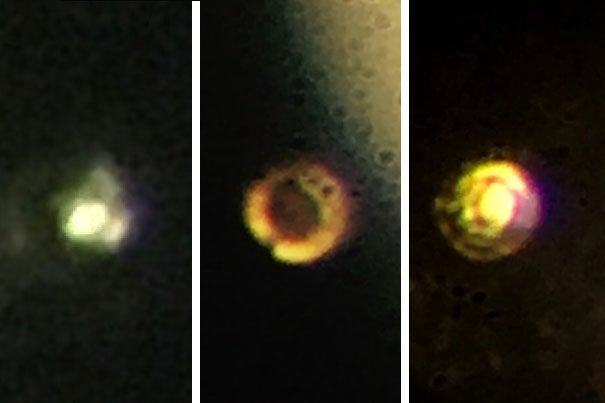
Thomas D. Cabot Professor of the Natural Sciences Isaac Silvera and postdoctoral fellow Ranga Dias decided that the best way to do this would be with diamonds.

Unfortunately even diamonds have defects, so after some trial and error the two realised they would need to take drastic action. They shaved five microns off the surface of the diamond and then coated it with an alumina to prevent the hydrogen from diffusing into the diamond.
They then squeezed a tiny sample of hydrogen at a staggering 71.7 million pounds per square inch. That’s more pressure than you’d find at the centre of the Earth.
It was under these incredible forces that the two noticed a change.
“It was really exciting,” said Silvera. “Ranga was running the experiment, and we thought we might get there, but when he called me and said, ‘The sample is shining,’ I went running down there, and it was metallic hydrogen.”
Of course it’s important to note that this paper has been met with more than a little healthy skepticism by its peers.
Speaking to the BBC, Eugene Gregoryanz from Edinburgh University called the paper “Complete garbage,” going on to say that, “Like everybody else who works with hydrogen at high pressures, I am appalled by what is being published in Science.”
Marcus Knudson from Sandia National Laboratories told the BBC that he had a more healthy outlook on the level of resistance to Harvard’s findings.
“The scepticism here is probably a good thing, in that it will drive many groups towards attempting to reproduce this experiment. This publication will certainly incite the field. Again, if it holds up, this is an exciting result. I think in this case time will tell,” he said.
Why is metallic hydrogen so important?
As explained by Silvera, metallic hydrogen could revolutionise a number of fields.
“It takes a tremendous amount of energy to make metallic hydrogen,” Silvera explained. “And if you convert it back to molecular hydrogen, all that energy is released, so that would make it the most powerful rocket propellant known to man, and could revolutionize rocketry.”
Then there’s metallic hydrogen’s role as a superconductor:
“As much as 15 percent of energy is lost to dissipation during transmission,” he said, “so if you could make wires from this material and use them in the electrical grid, it could change that story.”
