If you can remember any of your basic chemistry (we can’t) you will recall that helium holds a special place in the periodic table for being completely incapable of bonding with any other atom.
Well incredibly, it appears that this might not strictly be true, as a group of scientists claim to have done the complete opposite.

Remarkably, the team claim to have successfully bonded helium with sodium together.
This is huge news not least because it would prove wrong one of the most basic rules of chemistry but also because it gives us an incredible glimpse into the kind of chemistry that’s taking place in space.
You see there is only one other place where this ‘impossible’ bond could successfully take place: The centre of a gas giant.
To achieve the kind of conditions where helium could actually bond with another atom the team had to be quite inventive.
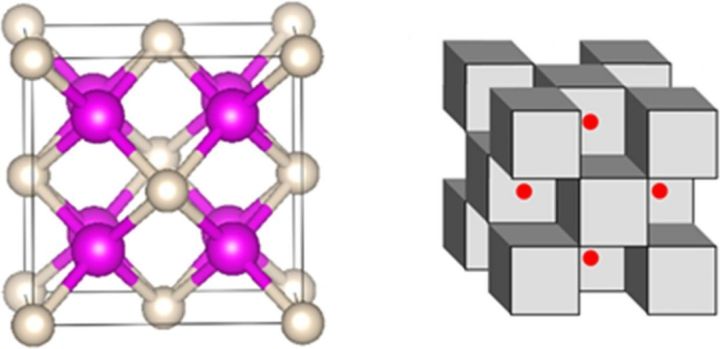
The researchers placed helium gas and sodium crystals in between two diamonds and then squeezed them placing unimaginable pressures onto the two atoms.
What they found at the end however was truly remarkable: A cube-shaped arrangement of alternating helium and sodium atoms with shared electrons.
Upon seeing these results Utah State University professor Alex Boldyrev then spent the nest two years along with his team just convincing fellow scientists and reviewers that this was in fact possible.
Finally the team were given the recognition that asked for and it poses big questions for what comes next.
It also raises big questions, Boldyrev says, that we must keep in mind when we consider the possibilities of humanity exploring beyond our own solar system./
“With the recent discovery of multiple exoplanets, we’re reminded of the vastness of the universe,” he says. “Our understanding of chemistry has to change and expand beyond the confines of our own planet.”
