Vietnam is the latest country to embrace a move towards ending animal testing of cosmetics. Last week the government confirmed that it will ban the Draize rabbit eye irritancy test for cosmetics purposes, paving the way for internationally accepted in vitro (non-animal) tests to be used instead.
The news comes as Humane Society International concludes its first Be Cruelty-Free Asia science tour, comprising high-level meetings and seminars with legislators, regulators and scientists in Hong Kong, China, Taiwan and Japan. The tour included a hands-on training programme for Chinese scientists which HSI funded with help from The Humane Society of the United States and the Human Toxicology Project Consortium. Experts from the Institute for In Vitro Sciences taught practical skills in how to conduct and interpret advanced non-animal test methods.

HSI and IIVS train Chinese scientists in non-animal tests for cosmetics
Vietnam is reported to be the only country in the Association of Southeast Asian Nations region to still conduct animal testing for cosmetics. Although the ASEAN Cosmetics Directive closely resembles the European Union's own cosmetics regulation, it doesn't yet contain the all-important animal testing ban. Before cosmetic companies can sell their products in Vietnam, they must first present safety data - including from animal tests - to the National Institute of Drug Quality Control. The IDQC also conducts random post-market animal testing checks. So HSI's Be Cruelty-Free campaign welcomes removal of the Draize eye test as a signal that Vietnam may be edging closer towards ending all cosmetics animal testing in future.
If it does so, it will join a growing number of countries across Asia and beyond that are embracing cruelty-free cosmetics. Following a high-profile Be Cruelty-Free campaign, India banned cosmetics animal testing in July last year, and this month proposed to ban the sale of animal-tested cosmetics too. Next month, China is set to remove mandatory animal testing for domestically-produced ordinary cosmetics, a regulatory change that HSI's team in Beijing has worked hard to help achieve. It's a clear signal that Chinese authorities are aware of and adapting to the global move away from decades-old animal test methods that simply can't keep pace with the high scientific standards of modern day regulatory requirements.

Legislators in South Korea sign the Be Cruelty-Free pledge
Australia, Brazil, New Zealand and the United States are all actively working with HSI or its affiliates and partners on legislative amendment proposals to ban cosmetics animal testing, and that's in addition to the testing (and sales) bans already in place across the 28 countries of the EU, as well as Norway and Israel.
In Taiwan, we're in deep negotiations with legislators to put forward a bill to ban cosmetics animal testing, as well as the sale of beauty products subject to new animal testing after the ban comes into force. Companies are under no legal obligation in Taiwan to conduct animal testing for 'regular' cosmetics, and have access to thousands of different ingredients from which they can manufacture safe new beauty products without any new animal testing, so we're working to make cruelty-free the only choice.
The justification often given for animal testing is that it allows you to test a substance in the whole organism. That's true, but it's simply the wrong organism. Mice and man diverged genetically about 75 million years ago. Although, like all mammals, we have a certain level of genetic similarity, this doesn't make mice 'little people' in the laboratory, and we should be extremely wary of attempts to extrapolate test results from one species to another.
Differences between humans and those animals typically used in cosmetics testing - rabbits, guinea pigs, mice and rats - include anatomy, metabolism, physiology and pharmacology. These differences are under-laid by genetic variations, which may be very subtle at the molecular level, but lead to significant organ or whole-body level consequences. Furthermore, test results vary not just by species, but even by different strains or genders within the same species. Far from being scientifically robust, using animals in 'safety' testing is often guess-work of the highest order.
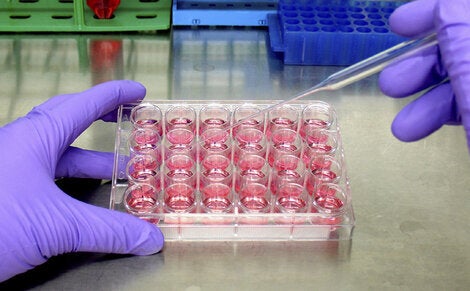
Cell culture test credit: ECVAM
By contrast, modern non-animal test methods have been scientifically validated to the highest international standards, and when used in combination with the thousands of existing ingredients already established as safe, they provide a faster, cheaper, more reliable and ethical way to assess cosmetics than animal testing.
The development of in vitro and other non-animal tests is a growing global business, due to be worth more than $17 billion by 2018. Perhaps unsurprisingly, Europe commands the largest share of the global in vitro toxicity testing market, and this has been attributed to the support provided by recent EU-wide legislation, specifically the 2009 cosmetics animal testing ban and the 2013 sales ban on newly animal-tested cosmetics. In technology-forward economies such as Japan, banning cosmetics animal testing would provide an exciting and lucrative opportunity to lead the world in next-generation techniques.
Some scientists in Japan are already engaged in developing state-of-the-art in vitro models. Researchers at Japan's National Institute of Agrobiological Sciences, for example, have developed an artificial human cornea called the Vitrigel-EIT method, which works by suspending a three-dimensional layer of human corneal cells on an ultra-thin collagen sheet. Toxicity is measured by counting the number of cells destroyed after application of a chemical. Although still in development, the research team has ambitions to apply the same approach to skin sensitisation, vascular permeability, and liver metabolism and toxicity.
Throughout our Be Cruelty-Free Asia science tour, the level of interest in cruelty-free cosmetics manufacturing has been striking. Without a doubt, many countries across the region are watching closely to see China implement its much-anticipated regulatory change. China's influence as a major Asian market is considerable, and our Be Cruelty-Free campaigns in Japan, South Korea, Taiwan and beyond have much to gain from China taking a step in the right direction.