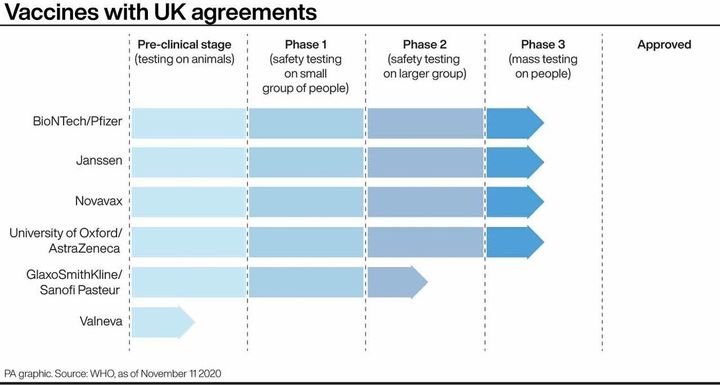
There are now four vaccines in the final stage of trials with UK agreements in place, which basically means we’ve agreed to buy millions of doses already.
The last stage of trials (phase three) involves each vaccine being issued on thousands of people to confirm firstly that it works, and secondly that there aren’t any adverse or dangerous side effects.
But what do we know about how these vaccines actually work? We’ll walk you through it.
BioNTech/Pfizer
This vaccine is called an mRNA vaccine – it uses a bit of the coronavirus’ genetic code to prompt the body to initiate an immune response.
While many standard vaccines work by injecting a dead or weakened form of a virus into the body in order to build immunity, RNA vaccines introduce a messenger RNA (mRNA) sequence into the body that contains the genetic instructions for the vaccinated person’s own cells to produce the vaccine antigens and generate an immune response.
It’s a bit like injecting an instruction manual so your body is able to build a bit of the virus protein, which then prompts an immune response. The body then knows how to fight the virus if you come into contact with it in real life.
Analysis of phase 3 trials has found it to be 95% effective against Covid-19, beginning 28 days after the first dose. To date, the Data Monitoring Committee for the study has not reported any serious safety concerns.
University of Oxford/AstraZeneca
This vaccine harnesses an adenovirus (common cold virus) from chimpanzees, which has been genetically modified so it can’t replicate in our body’s cells. This virus is used as it contains the genetic material of the SARS-CoV-2 virus spike protein (a protein that lives on the surface of the coronavirus).
When the vaccine enters cells inside the body, it uses this genetic code to produce the surface spike protein of the coronavirus, priming the immune system to attack the SARS-CoV-2 virus if it later infects the body.
Analysis of phase 3 trials has found it to be 70.4% effective when combining data from two dosing regimens. In two different dose regimens, vaccine efficacy was 90% in one and 62% in the other. The more effective regimen used a halved first dose and standard second dose. No serious safety events related to the vaccine were identified.
Professor Andrew Pollard, director of the Oxford Vaccine Group and chief investigator of the Oxford Vaccine Trial, said: “Excitingly, we’ve found that one of our dosing regimens may be around 90% effective and if this dosing regimen is used, more people could be vaccinated with planned vaccine supply.”
Janssen
The vaccine is an adenoviral vaccine, so it’s very similar to the University of Oxford/Astrazeneca vaccine in terms of how it works. However it’s different in that the adenovirus used is from humans (rather than chimpanzees). The virus has also been modified, so it can’t multiply in the body or cause illness.
This vaccine is currently in phase 3 trials.
Novavax
The Novavax vaccine uses a slightly different technology to the others – it is engineered from the genetic sequence of SARS-CoV-2. The company’s technology turns insect cells into factories for the coronavirus spike protein and the vaccine combines several of the spike proteins in a nanoparticle.
It also contains something called an adjuvant, which is used in other vaccines to enhance the immune response and stimulate high levels of neutralising antibodies. The UK already provides an adjuvanted flu vaccine to the over-65s, so we know this technology is safe and works.
This vaccine is currently in phase 3 trials.