A groundbreaking new treatment that actually alters the immune system of someone with cancer has been given FDA approval in the US.
Known as gene therapy, the treatment involves removing immune cells called T cells from the patient’s blood and then genetically altering them in a lab.
They insert a gene for a protein called a chimeric antigen receptor which is effectively a set of instructions for it to start attacking leukaemia cells.
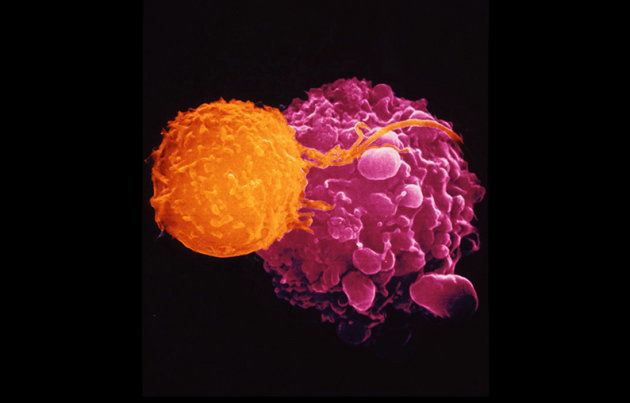
The modified cells are then transfused back into the patient.
Called Kymriah, the treatment has been designed to attack a very specific type of blood and bone marrow cancer that effects children and young adults.
Novartis, the pharmaceutical company behind the treatment, saw complete remission in 83% of 63 patients who had B cell acute lymphoblastic leukemia.
Gene therapy has been heralded as a powerful new weapon in the fight against cancer with some experts even claiming that eventually it could signal the end of the disease.
That’s a long way off however, and for now the treatment isn’t without its risks.
In some cases what you’re effectively doing is removing the ‘brakes’ on the body’s immune system, a decision that can come with some fairly brutal side effects.
Novartis is no exception with side effects ranging from high fevers, diarrhoea and vomiting to even delirium, loss of balance and difficulty speaking and understanding.
Then of course there’s the difficulty in actually administering the treatment. Currently, gene therapy is carried out on an individual basis with each person having their cells sent to the lab for alteration.
This involves having to travel to a limited number of approved centres across the USA and then waiting for the individually tailored treatment to be sent back.
Finally there’s the cost, and in this case it’s high. Novartis say the drug will cost $475,000. That’s a lot.

To try and alleviate fears over the cost the company is reportedly looking into a scheme where it won’t charge the government or the insurance company unless the treatment has shown to be effective after a month.
Despite these negatives, FDA approval of gene therapy is a big step for the treatment both in the USA and around the globe.
Commenting on the decision, FDA commissioner Scott Gottlieb said: “We’re entering a new frontier in medical innovation with the ability to reprogram a patient’s own cells to attack a deadly cancer.”
“New technologies such as gene and cell therapies hold out the potential to transform medicine and create an inflection point in our ability to treat and even cure many intractable illnesses.”
