A groundbreaking gene therapy treatment that alters a patient’s own blood cells into targeting cancer cells has seen some remarkable results.
Of those tested, a third of patients suffering from extremely advanced lymphoma are still showing no signs of the disease a full six months after having just a single treatment.
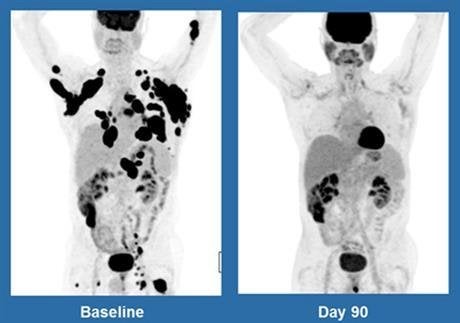
The study, which was sponsored by the pharmaceutical giant Kite Pharma, has resulted in 36 per cent of patients in the trial going into complete remission.
What makes this even more promising is that six months later this figure has barely changed, suggesting that this treatment could be a powerful new alternative when no other treatments have succeeded.
“These results with axicabtagene ciloleucel are exceptional,” said Jeff Wiezorek, M.D., Senior Vice President of Clinical Development, “and suggest that more than a third of patients with refractory aggressive NHL could potentially be cured after a single infusion of axicabtagene ciloleucel,”
The treatment isn’t without risk however, gene therapy of this type involves radically altering your own cells to remove the conventional ‘brakes’ that stop them from attacking cancerous cells.
It’s a brutal approach signified by the fact that of the 101 who participated two are believed to have died from the treatment itself, not their cancer.
Overall though, the signs have been extremely promising. Six months after treatment 41 per cent have found that their cancer shrunk at least by half.
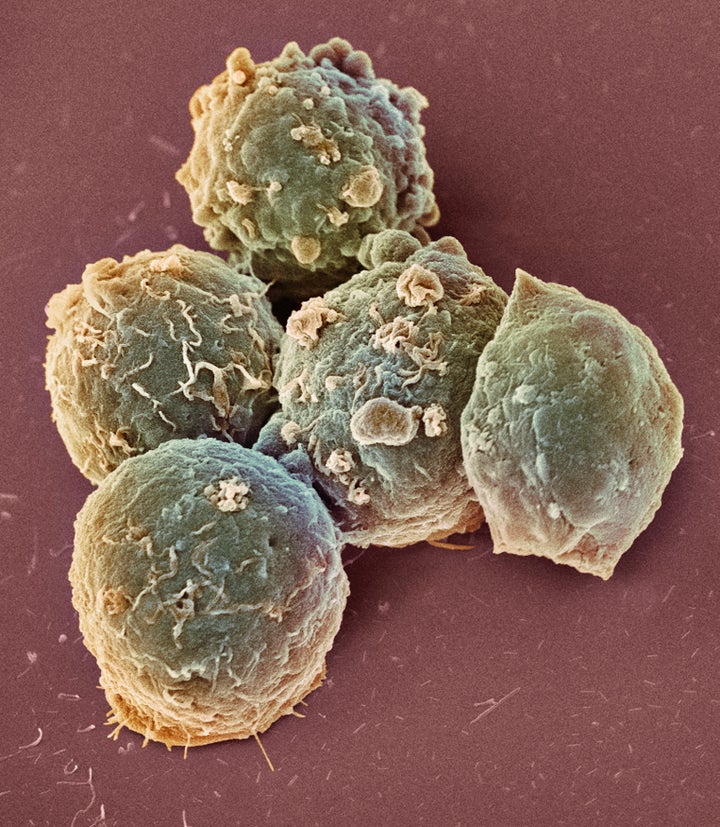
The treatment itself involves filtering a person’s blood so that they can remove a key component of the immune system called T-Cells and then modify them in a laboratory.
They add a gene to the T-Cells that will force them to target cancer cells and then the blood is injected back into the person intravenously.
Once inside the body these altered cells multiply in huge numbers and are able to attack the cancer.
Speaking to AP Dr. Fred Locke, a blood cancer expert at Moffitt Cancer Center in Tampa said “The numbers are fantastic, these are heavily treated patients who have no other options.”
The next step will be gain full approval for the treatment from the U.S. Food and Drug Administration later in March and then from Europe by the end of the year.
