When your body is targeted by a dangerous infection, the army of cells responsible for ensuring you can fight it off and return to health, are your T-cells.
In most cases the immune system deploys these T-cells as a primary weapon and they are able to kill off the foreign bodies effectively, but not when it comes to cancer.
This is because cancer cells are smart enough to evade detection - a problem that cancer scientists are trying to fix by engineering designer T-cells that have a ‘turbo’ functionality.
In many previous studies researchers have successfully engineered synthetic T-cells to combat tumours, but the resulting cells were not ideal as they had significant side effects and were really technically challenging to create.
Now a team from the Department of Biosystems Science and Engineering in Basel have come up with a simpler way to make “innovative” designer cells.
Professor Fussenegger said: “Our innovative T-cells may detect and kill metastasising cancer cells at a very early stage, when other treatments are not effective.”
Using renal cells and adipose stem cells, Fussenegger and his colleagues have created ‘fake’ T-cells that mimic their behaviour with three added features.
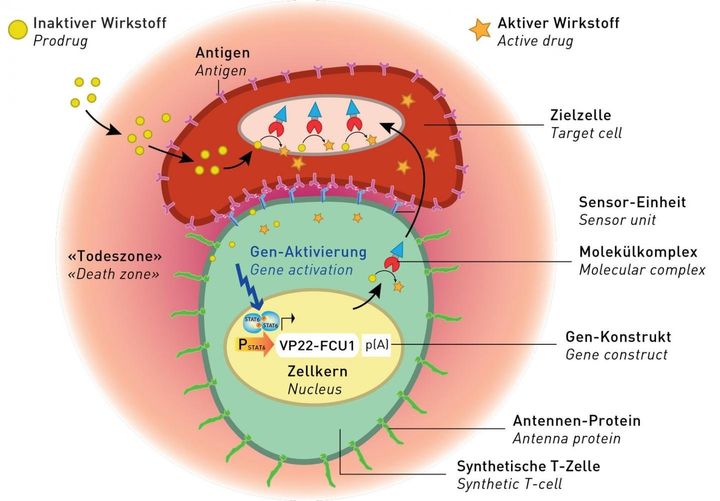
Firstly they have molecular antennae protruding well outside the membrane, which buckle to the cancer cells and allow it to anchor itself inside.
Secondly, it has antibodies inside cells with docking sites that can sense cancer and bind to it, and thirdly they have a molecular ‘warhead’ that penetrates the membrane of the target cell.
The cancer cells then absorb this substance, and the converter module transforms it from an inactive to inactive state causing the cancer cells to burst.
Not only does this destroy the initial cancer cell, but as it bursts, re-releases the active substance and destroys other tumour cells in the “death zone” surrounding the cell.
This therapy would have several advantages over current treatments: “The T-cells operate totally independently from the body’s immune system, enabling it to continue to function perfectly normally, so that fewer side-effects are likely,” says Fussenegger.
During chemotherapy the body is flooded with active substances in order to kill as many rapidly dividing cells as possible in an unselective manner, only a few artificial T-cells are needed in the new therapy, and these are deployed in a targeted fashion.
So far, researchers have only tested their new cells in cell cultures and it is not clear how the system will function in the human body.
Fussenegger said: “At present our new system is still a long way from a therapeutic application.”
