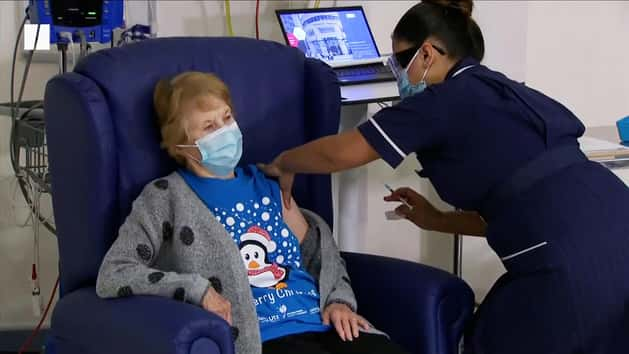
A 90-year-old has become the world’s first patient to receive the Pfizer/BioNTech Covid-19 vaccine.
Margaret Keenan received the jab at Coventry Hospital on Tuesday morning, as dozens of hospital hubs across the country gear up to deliver millions of vaccines.
The vaccine produced by Pfizer and German biotech company BioNTech has made headlines recently after it was approved for use in the UK.
The company expects to make as many as 50 million vaccine doses this year, enough to protect 25 million people, and then produce up to 1.3 billion doses in 2021.
So what do you need to know about the vaccine? We’ll walk you through what we’ve learned so far.

1. How it works
It’s an mRNA vaccine, so it uses a bit of the coronavirus’ genetic code to prompt the body to initiate an immune response.
While many standard vaccines work by injecting a dead or weakened form of a virus into the body in order to build immunity, RNA vaccines introduce a messenger RNA (mRNA) sequence into the body that contains the genetic instructions for the vaccinated person’s own cells to produce the vaccine antigens and generate an immune response.
It’s a bit like injecting an instruction manual so your body is able to build a bit of the virus protein, which then prompts an immune response. The body then knows how to fight the virus if you come into contact with it in real life. Clever.
The benefit of this particular vaccine is because the RNA is man-made, you don’t need live virus to make a batch of the vaccine. This makes it generally easier to manufacture.
2. Who came up with the idea?
A married couple of Turkish descent are the brains behind the vaccine. Professor Ugur Sahin, 55, and Dr Ozlem Tureci, 53, run German biotech company BioNTech.
Their company primarily specialises in individualised cancer medicine, but in January they pivoted to creating mRNA-based viral vaccines to help fight Covid-19, Sky News reported.
3. How effective is the vaccine?
Data from the Phase 3 clinical trial demonstrated a vaccine efficacy rate of 95%. Efficacy was consistent across age, gender, race and ethnicity demographics, with an observed efficacy in adults age 65 and over of more than 94%.
The vaccine is given in two doses, and protection from the vaccine is achieved 28 days after the first injection. The protection that the vaccine offers is that it can help prevent people from becoming severely ill and needing hospitalisation because of the virus. At this stage we don’t know if it can prevent actual transmission of the virus.
4. Is the Pfizer vaccine safe?
The Pfizer vaccine has been approved for use in the UK by the Medicines and Healthcare products Regulatory Agency (MHRA) which means it’s been deemed safe to use for those aged 16 and over. Further tests are required to determine whether it’s safe for children and pregnant women.
5. Are there any downsides?
One of the biggest issues is that the vaccine needs to be stored at an incredibly cold temperature in order to work (-80 °C to -60 °C), which brings distribution challenges.
Some people may experience side effects from having the vaccine, however this can occur with most vaccines so it’s nothing new. Some of the more common side effects, which may affect one in 10 people, include: pain at the injection site, tiredness, headache, muscle pain, chills, joint pain and fever. People are urged to tell their doctor, pharmacist or nurse if they experience any side effects.
6. What happens to other vaccines now?
Trials of other vaccines are still going ahead around the world – based on results of trials so far, it’s likely there’ll be a few approved vaccines circulating by this time next year.